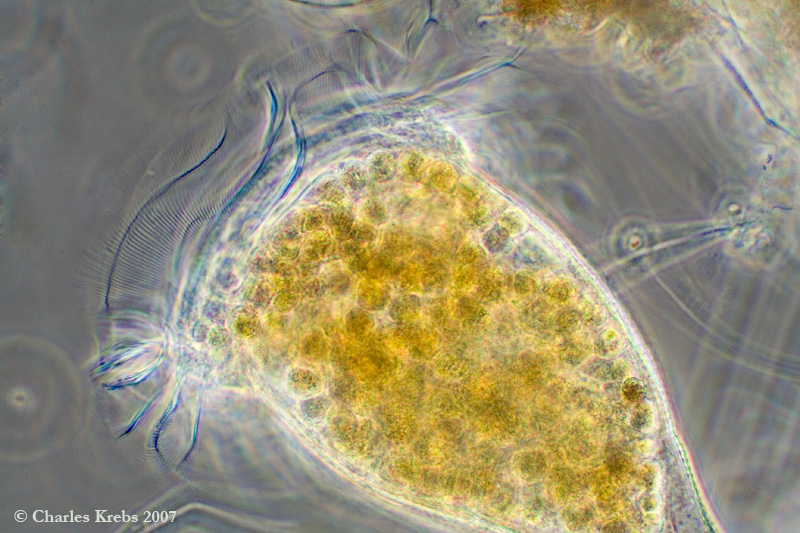
Dominique... good to hear from you. (You need to post more pictures

)
While I do not have a photo of the completed setup, I do have one picture I made while it was "a-work-in-progress". But this photo illustrates the important features, and it can be seen here:
http://krebsmicro.com/forumpix/BHS_flash.jpg
On the Olympus BHS, the entire bulb/condenser "unit" simply plugs into the rear of the stand. I was fortunate to locate a second complete lighting unit and make the modifications to one as seen in this picture. The picture referenced shows the "stock" BHS 100W bulb and collector on the left side. On the right is the modified unit. I positioned a flash tube at the exact position formerly occupied by bulb. The arrangement that holds the flash tube permits it to be adjusted forward and back, as well as up and down. It can be positioned very precisely to maximize the light that passes through the "collector" condenser. After this picture was taken I added a second "platform" directly behind the flash tube that holds a Luxeon K2 LED. This way I am able to fully adjust the position of the flash and LED. The LED is there to provide my viewing and focusing light.
The flash unit I used was one of my Vivitar 283's with the control "box" that was discussed in the "old" forum (There is also a page on my Krebsmicro.com site). This time, the flash tube was removed from the flash. Every time disassembling a small electronic flash unit is discussed the following is very important to remember:
Extreme caution must be used since the power stored in the capacitors of these flashes can be dangerous! The shock hazard is very real. All capacitors must be properly, and completely, discharged before doing any work.
Gerd Günther did an nice internal modification like this to a Leica stand, but the page he posted describing it does not seem to exist any longer.
This is the 4th method of incorporating flash that I have tried and it provides the most light. I was fortunate because the BHS light assembly made it fairly easy to accomplish. When I first started using the BHS I had set up a 70/30 pellicle mirror at a 45 degree angle just above the light port. The flash was directed into that... and the method actually worked very well once aligned properly. I would probably have developed that method and still be using it if it had not been so easy to make the modification to a BHS assembly.