Shiny! The insides of a wasp mouth...
Moderators: rjlittlefield, ChrisR, Chris S., Pau
You get smaller crystals if you cool things quickly, whether freezing or by precipitation, so it might be good to do that somewhere aloing the line.
Some chems are far more soluble in warm water than cold, the cooling would initiate faster precipitation in those.
If I remember correctly, NaCl doesn't dissolve much better in warm water than cold. In fact KCL dissolves better in cold than hot - unusual or I'd never have remembered. Low molecular weight sounds good, eg not a sugar, for small molecules/crystals.
I've searched a little for clues for what might form small , regular crystals, but all the documents I find are all about growing great big ones.(When I was a lad a silicon crystal for semiconductors was amazingly big at 20mm, but Andrew pointed out it's now it's more like 300mm!)
Another approach you could try would be a monolayer of a fat molecule.
Methanol or something more pure than the Bitrex/purple stained stuff, like Vodka, with a tiny amount of cooking oil added. Almost none. The fat molecules would repel each other but wind up aligned, more or less, on the surface when the solvent evaporated.
ANother thought - a dye, like Fluorescein or maybe one of the standard micrographic ones. I have some drain-testing dyes I could try.
Some chems are far more soluble in warm water than cold, the cooling would initiate faster precipitation in those.
If I remember correctly, NaCl doesn't dissolve much better in warm water than cold. In fact KCL dissolves better in cold than hot - unusual or I'd never have remembered. Low molecular weight sounds good, eg not a sugar, for small molecules/crystals.
I've searched a little for clues for what might form small , regular crystals, but all the documents I find are all about growing great big ones.(When I was a lad a silicon crystal for semiconductors was amazingly big at 20mm, but Andrew pointed out it's now it's more like 300mm!)
Another approach you could try would be a monolayer of a fat molecule.
Methanol or something more pure than the Bitrex/purple stained stuff, like Vodka, with a tiny amount of cooking oil added. Almost none. The fat molecules would repel each other but wind up aligned, more or less, on the surface when the solvent evaporated.
ANother thought - a dye, like Fluorescein or maybe one of the standard micrographic ones. I have some drain-testing dyes I could try.
-
gpmatthews
- Posts: 1040
- Joined: Thu Aug 03, 2006 10:54 am
- Location: Horsham, W. Sussex, UK
- Contact:
You could try a film coat as a means of altering surface texture. A simple formulation would be something like hydroxypropylmethylcellulose (HPMC - basically wallpaper paste) with up to 1% or so high molecular weight polyethylene glycol (PEG 4000 or PEG 6000, the number is the approx MW) as plasticiser and a trace of detergent as wetting agent all dispersed and applied in water (use less than ~10% solids) and air dried. HPMC could also be used to poison crystal growth so that you prevent formation of large crystals if trying out a fine crystalline coating. HPMC coats are likely to be matt and of lower reflectivity than glossy insect chitin. Other cellulose derivatives, such as methylcellulose will probably work as well as HPMC. HPMC 3 cps (3 cps is the viscosity of a standard concentration dispersion) is commonly used in film coats on tablets. To disperse cellulose derivatives successfully in water, mix with very hot water and stir continually whilst cooling, this minimises lumps which form in cold dispersions (these compounds are generally insoluble in hot water). You can add solids such as talc to film coating supensions to alter the properties of the dry film.
Being water soluble, you can wash coats off and try again if not successful.
Here is a whole new area for experimentation!
Being water soluble, you can wash coats off and try again if not successful.
Here is a whole new area for experimentation!
Graham
Though we lean upon the same balustrade, the colours of the mountain are different.
Though we lean upon the same balustrade, the colours of the mountain are different.
I wonder how big "flowers of sulfur/sulphur" are. Or corn starch powder.
I also wonder how fine is the stuff the women stick on their face. Looking though a 40x lens, jolly fine. It seems the more you pay, the finer it gets.
Some numbers:
http://www.shreejifinechem.com/talc_powd.htm
http://www.cxchuangxin.com/pages/talcum-powder.htm
and on this page under mesh sizes there's an interesting suggestion for a 2 micron particle...
http://www.skylighter.com/fireworks/mak ... e-size.asp
I also wonder how fine is the stuff the women stick on their face. Looking though a 40x lens, jolly fine. It seems the more you pay, the finer it gets.
Some numbers:
http://www.shreejifinechem.com/talc_powd.htm
http://www.cxchuangxin.com/pages/talcum-powder.htm
and on this page under mesh sizes there's an interesting suggestion for a 2 micron particle...
http://www.skylighter.com/fireworks/mak ... e-size.asp
- rjlittlefield
- Site Admin
- Posts: 23608
- Joined: Tue Aug 01, 2006 8:34 am
- Location: Richland, Washington State, USA
- Contact:
-
Peter M. Macdonald
- Posts: 187
- Joined: Tue Jan 20, 2009 2:59 pm
- Location: Berwickshire, Scotland
Well there's always a
sublime answer:
You really want something which de-sublimes (is there a word for that?) straight from vapour to solid, a bit like your headlamp filament tungsten, but at say 40ºC!
Vapour deposition does tend to produce shiny surfaces, of course like on camera lenses.
like on camera lenses.
I know industrially they charge things to help the process, like Andrew's needle in reverse. Fine white smoke then? Magnesium? MgO is a small molecule...
sublime answer:
You really want something which de-sublimes (is there a word for that?) straight from vapour to solid, a bit like your headlamp filament tungsten, but at say 40ºC!
Vapour deposition does tend to produce shiny surfaces, of course
I know industrially they charge things to help the process, like Andrew's needle in reverse. Fine white smoke then? Magnesium? MgO is a small molecule...
- rjlittlefield
- Site Admin
- Posts: 23608
- Joined: Tue Aug 01, 2006 8:34 am
- Location: Richland, Washington State, USA
- Contact:
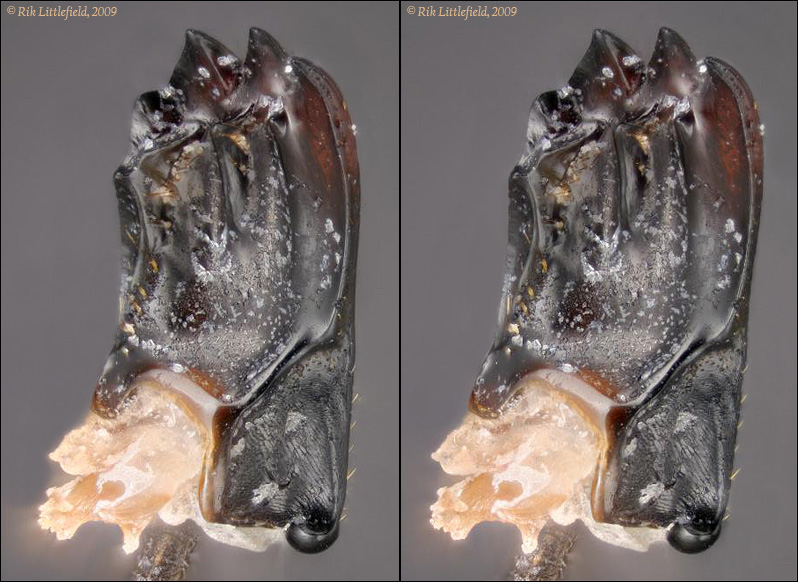
No change to the coating here, just a different position of the mandible.
I think this works pretty well to illustrate the structure.
I've also included the base of the mandible, since we can see some of the functional anatomy really well in this view. Notice in particular the massive ball joint at the bottom right hand side, and the remains of that huge muscle bundle coming out of the base. These critters may have a little trouble getting their mouths open, but getting them shut again is no problem at all!
105 frames, 0.00033 inches focus step, ZS PMax with mild retouching.
--Rik
